Regulation Catalogue
Find specific regulations without interference
Finding the specific regulation you need in a hurry can be difficult. Government institutions, news outlets and the scientific community produce such a volume of material that you can easily be overwhelmed and distracted.
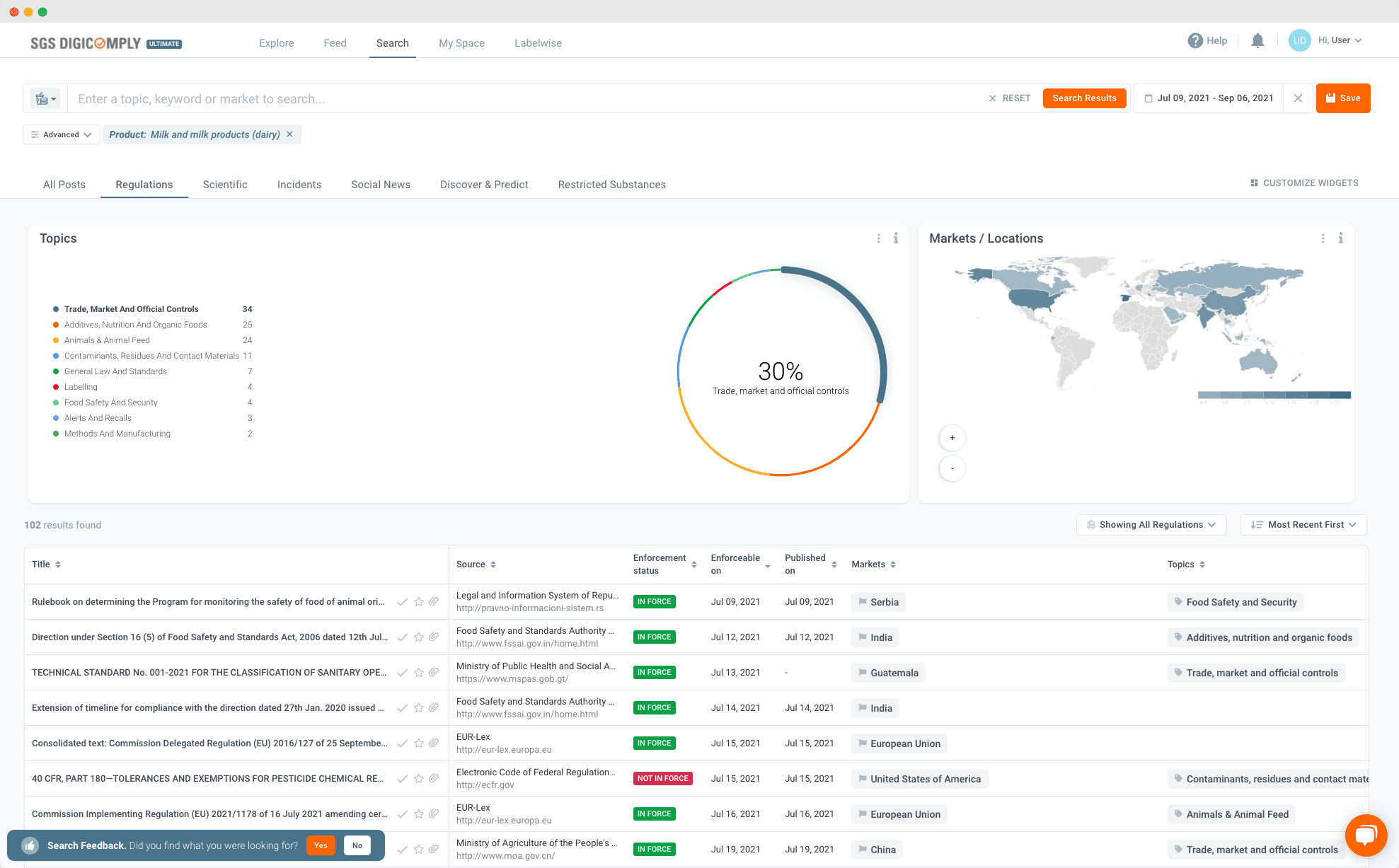
We developed Regulation Catalog to answer the call from SGS DIGICOMPLY users for a better and quicker way to find regulations. Our innovative search function lets you distil down the legislation affecting food companies, retailers and personal care product companies. It separates the facts from the ‘noise’, using keywords to search and deliver results that can be further filtered by market, product category, topic and source. This system also allows the quick identification of in force dates and related amendments.
This solution is ideal for the following industries:
-
 Food Manufacturer
Food Manufacturer -
 Food Service & Distribution
Food Service & Distribution -
 Retailer (including online)
Retailer (including online) -
 Agricultural & Feed Producer
Agricultural & Feed Producer -
 Pet Food Manufacturer
Pet Food Manufacturer -
 Food Supplements
Food Supplements

Why is it detecting food safety risks so hard?
Read ArticleSpotlight on Solutions


The most advanced data-driven platform for product risks and opportunities.
Learn More
Millions of continuously updated insights for food-related risks.
Learn More
Leverage the consumer’s voice to gather food safety risk intelligence.
Learn More
A comprehensive database of food and non food product ingredient limits
Learn More
Find specific regulations among millions of documents without interference.
Learn More
Where to start when navigating a country’s regulatory environment with a trusted source.
Learn More



Validate labels and the content in your product’s artwork with Labelwise.
Learn More


A wealth of food safety and compliance data at your disposal for API and services integration.
Learn More
