New SGS DIGICOMPLY Features for 2022 Simplify Compliance and Risk Reduction
There is a well-known phrase in business – time is money. SGS DIGICOMPLY – the artificial intelligence driven data gathering solution for risk prediction and regulatory compliance – has always helped operators in the food and feed and cosmetics, personal care and household (CPCH) sectors to save time and money. With the launch of three new features as part of our 2022 Spring Release, we have expanded our offering and stakeholders are now able to achieve even greater efficiencies in their value chain.
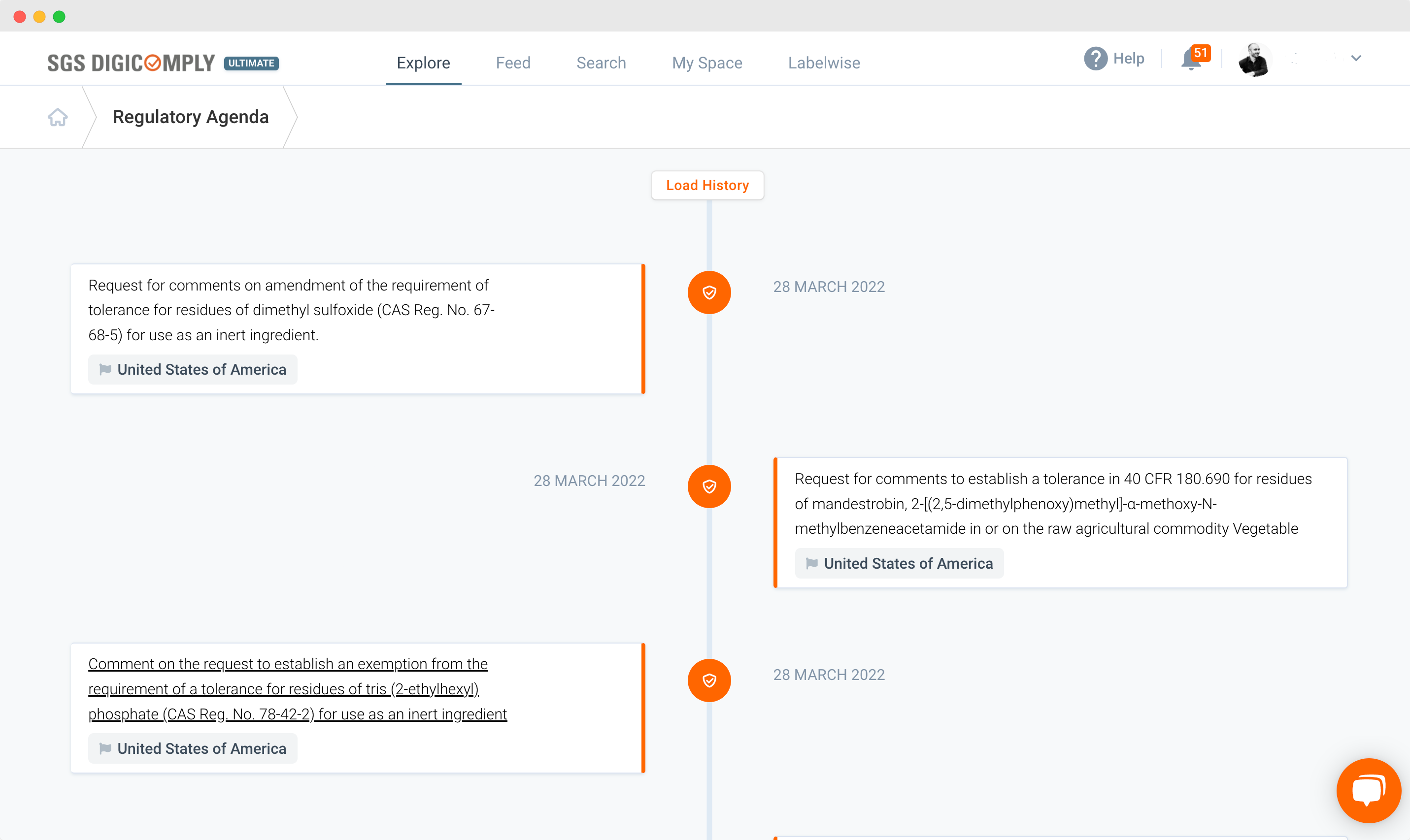
Introducing Regulatory Agenda – instant access to business-critical information
Operators in the food and feed sectors know how quickly the regulatory landscape can change. Staying up to date with changes that can impact products and supply chains can be difficult. It can be challenging to find, assimilate and action relevant legislative updates, leaving businesses exposed.
Regulatory Agenda is a versatile tool that provides users with an interactive snapshot of all relevant regulatory changes that may impact their business.
Users simply log into SGS DIGICOMPLY, choose ‘Explore’, and then click on ‘Regulatory Agenda’. They are then transported to a single page that contains a timeline of regulatory updates – most recent first. It displays only updates that are relevant to the user’s profile, color-coding them for ease of navigation – orange meaning the update has happened and blue meaning it is forecast.
Regulatory Agenda offers quick and easy access to all relevant documentation. In addition to regulatory updates, users will also find links to discussion papers, consultation documents, interim and final reports, and communications for industry bodies and other stakeholders. Through one portal, they can efficiently access all relevant documents, enabling them to remain compliant in an ever-changing regulatory landscape.
Introducing Dossier Dashboard – data and documents at a glance
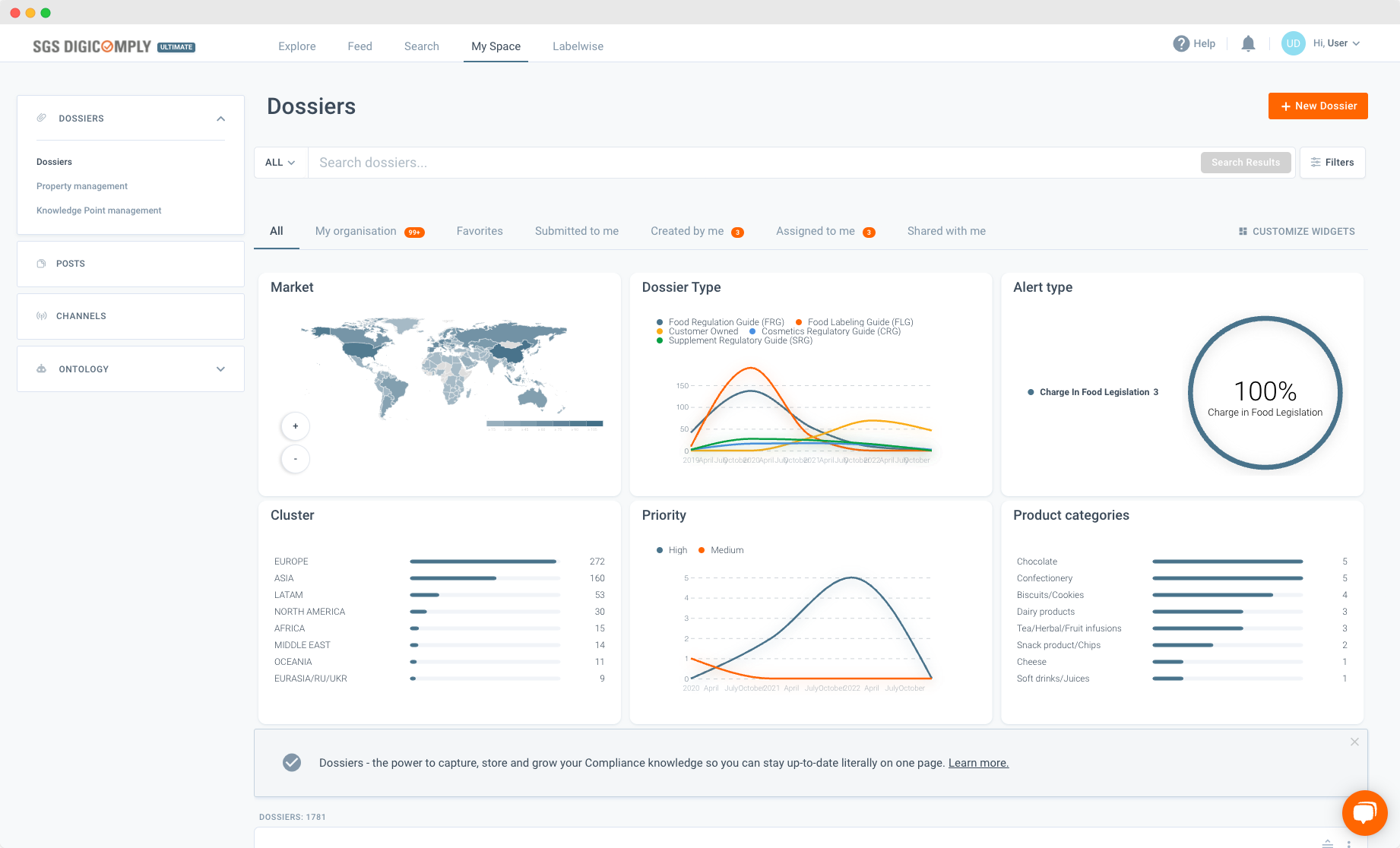
SGS DIGICOMPLY has always offered users the ability to collect relevant documents in a single electronic file – a dossier. Each dossier will contain aggregated knowledge drawn from the millions of documents available on the SGS DIGICOMOPLY platform. However, the complexity of this data meant it could be difficult to visualize.
Dossier Dashboard enriches the user’s experience. Within the dossier, they can now gather relevant information and then create a dynamic visual representation that enables a rapid overview of pre-determined data.
This user-friendly feature empowers the visual summarization of content by factors such as:
- Market (geographical market)
- Dossier type (the type of file within the dossier – food regulatory guides, food labelling guides, customer owned files, cosmetics reg. guide, supplementary reg. guide)
- Alert type
- Priority (high, medium, low)
- Product categories (e.g., Chocolate, Confectionery, Biscuits, etc.)
- Cluster (geographical groupings – Europe, Asia, LATAM, North America, Middle East, Oceania, Eurasia/RU/UKR)
Introducing Compliance Request Portal – an innovative way to access regulatory professionals
Whether a company sells a single product to a single market or is a large multinational delivering a variety of products to multiple markets, compliance matters. The boundary between human and digital is blurring and so, with more and more regulatory authorities and service providers now operating online, it is no longer enough to rely on an offline approach to compliance and risk prevention.
Compliance Request Portal bridges the gap, offering a completely managed and delivered regulatory advisory service to clients of all sizes. The portal gives users direct access to SGS DIGICOMPLY’s network of regulatory analysts.
Depending on their requirements, analysts can provide:
- Customized specialist expertise based on geography, regulatory knowledge and language skills
- Support – from small research projects to complete outsourcing
- Interpretation and summarization of research findings
- Reviews of product specifications; nutritional, health and marketing claims; market entry requirements
Clients submit a ‘Technical Advisory Request’ via the portal. This is then reviewed by SGS DIGICOMPLY experts, who interpret the requirements and devise a solution with quote. Upon payment, analysts will create an online dossier with live links to relevant regulations (in English). Because this is a ‘live’ dossier, the client will then be notified every time these is an update, with links to the relevant documents in the original language and an English translation.
Compliance Request Portal goes beyond the traditional DIGICOMPLY report; it is a single place of contact for:
- Submitting, approving and tracking requests
- Communicating directly with SGS DIGICOMPLY analysts
- Accessing all current and past dossiers
Looking forward
When noncompliance isn’t an option and time is money, SGS DIGICOMPLY is the first choice for dynamic intelligence and effective risk management. With the introduction of Regulatory Agenda, Dossier Dashboard and the Compliance Request Platform as part of the 2022 Spring Release, SGS DIGICOMPLY achieves greater customer-centric utility, with features that enable rapid assessment and response to changes in the compliance landscape.


.webp?width=1644&height=1254&name=Food%20Safety%20Dashboard%201%20(1).webp)
